Water in oil emulsion volum fraction9/27/2023 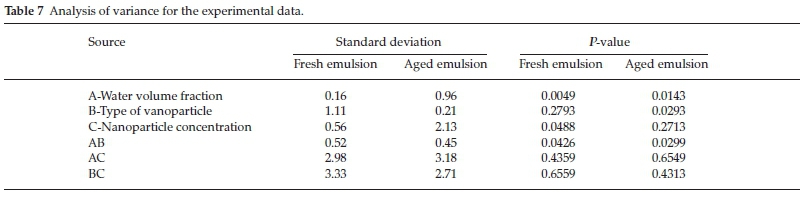
In addition, the natural polarity of asphaltenes is capable of fortifying the emulsion stability ( Dehghan et al., 2013). (1992) have concluded that the majority of carboxylic acid groups in the heavy oil is made up of asphaltenes, phenols, or resins. Thus, the total acid number (TAN) of oil becomes an important index that implies the feasibility of alkaline flooding in a specific reservoir. Different from in surfactant flooding, the alkaline solution reacts with the acidic groups of residual oil through neutralization reactions and generates in situ surfactants spontaneously ( Rivas et al., 1997 Sun et al., 2008 Ashrafizadeh et al., 2012). These goals can be achieved by incorporating the solvent injection, thermal treatment, and chemical injection to efficiently recover an additional 10% original oil in place (OOIP) or more from the reservoir ( Kokal and Abdulaziz, 2010).Īs one of the chemical EOR processes, alkaline flooding has numerous merits which are mainly represented by its low cost ( Ding et al., 2010) and high versatility. 
Most of the EOR processes focus on scaling down the mobility ratio to further diminish the S or. In order to recover more oil, enhanced oil recovery (EOR) is implemented following water flooding. Severe viscous fingering contributes to early water breakthrough and high residual oil saturation (S or) ( Homsy, 1987 Bryan and Kantzas, 2007). Nonetheless, the low viscous water that displaces fluid results in an unfavorable mobility ratio. Water flooding as a secondary recovery method is conducted afterward to maintain the reservoir pressure and mobilize the initially unmovable oil. However, the primary recovery is not long-lasting in the heavy oil reservoir due to the rapid reservoir pressure depletion and high viscous nature of the heavy oil. Initially, the primary production utilizes pressure depletion owing to formation rock expansion and solution gas drive to make oil produced spontaneously ( Kokal and Abdulaziz, 2010). The heavy oil recovery process in field mainly undergoes three stages. The high versatility of the modified bottle test ensures that the alkali usage is not limited to the inorganic alkalis mentioned in this study other type of alkaline solutions can also be used for further expanding the scope of its application. The statistically optimized experimental designs were implemented due to the simplicity of the new bottle test method and it considerably cut the time expense regarding the alkaline flooding performance prediction. It is found that the majority of emulsion effluent type from the sandpack flooding test were in agreement with the bottle test forecast which proved the feasibility of the modified bottle test method. Afterward, material balance calculations other than pure volume observation were applied to quantify the emulsion volume and determine the major emulsion type formation. To accurately evaluate the emulsion type and phase volume distribution from the bottle test, each emulsion phase after aging in the test bottle was sampled and its water content was measured through Karl Fischer titration. The modified method considers the necessary energy input required for mixing immiscible bulk phases at low interfacial tension (IFT) regions to improve the representativity of emulsion formation in the bottle test to that of in porous media. In this study, a modified bottle test method that assesses major emulsion type formation for preliminary prediction of alkaline flooding performance in oil recovery is introduced. Program of Petroleum Systems Engineering, Faculty of Engineering and Applied Science, University of Regina, Regina, SK, Canadaĭue to the diversity of alkali categories and reservoir conditions, the varied oil recovery driving mechanism of alkaline flooding is subjected to different types of emulsion generation.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
